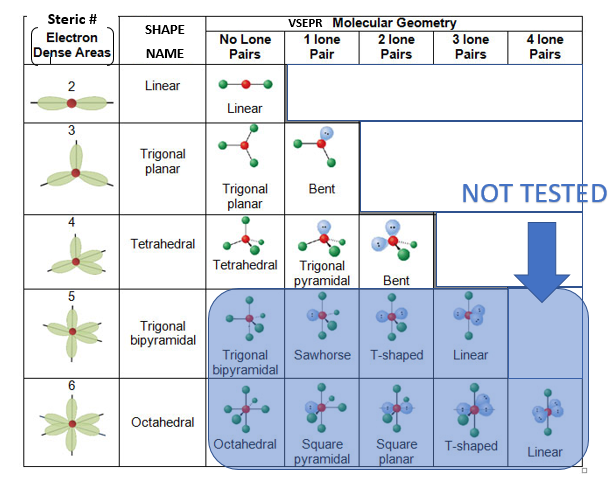
| chapter_4_clicker_review.pptx | |
| File Size: | 698 kb |
| File Type: | pptx |
HELPFUL CLIMATE LINKS...(videos from a filmmaker friend of mine)
- https://www.youtube.com/watch?v=vhyau8xRIfQ
- https://www.youtube.com/watch?v=xpNSpNBVksQ
https://www.youtube.com/watch?v=fYzk2YgxoiI
https://www.youtube.com/watch?v=vX3drC8G21o
ALL STORYBOARDS WILL BE PRESENTED IN LIBRARY THEATER ON MON 2/3!
Example of a semester 1 graphic organizer
Chapter 12 "I CANs"...
...use periodic trends to predict relative atomic radius, ionization energy and electronegativity.
...explain changes in size when an atom becomes either a cation or anion.
...distinguish between ionic, polar covalent and nonpolar covalent bonds & explain how they are formed.
...characterize the polarity of both bonds &/or molecules. Draw dipole moments on 3D VSEPR sketches of polar molecules.
...create proper Lewis structures for simple molecules.
...apply VSEPR theory to predeict the shape (molecular structure), VSEPR label & bond angle of simple molecules from their Lewis structures.
...use periodic trends to predict relative atomic radius, ionization energy and electronegativity.
...explain changes in size when an atom becomes either a cation or anion.
...distinguish between ionic, polar covalent and nonpolar covalent bonds & explain how they are formed.
...characterize the polarity of both bonds &/or molecules. Draw dipole moments on 3D VSEPR sketches of polar molecules.
...create proper Lewis structures for simple molecules.
...apply VSEPR theory to predeict the shape (molecular structure), VSEPR label & bond angle of simple molecules from their Lewis structures.
| bonding_review.pptx | |
| File Size: | 262 kb |
| File Type: | pptx |
| lewis_structure_lab.docx | |
| File Size: | 16 kb |
| File Type: | docx |
| periodic_trends.ppt | |
| File Size: | 535 kb |
| File Type: | ppt |
Chapter 11 "I CANs"...
...express electron configurations in orbital box diagrams and noble gas notations (including Cr & Cu exceptions)
...compare and contrast the Bohr model of the atom (orbits) with the wave mechanical model (orbitals).
...visualize and explain orbital shapes (sublevels)
...state possible #'s of electrons in various orbitals, sublevels and principle energy levels.
...determine the number of valence electrons in an atom.
...locate the s,p,d & f blocks on the periodic table.
...explain and diagram major wave vocab (amplitude, speed, wavelength, frequency, Hertz & energy)
...compare/contrast relative speed, wavelength, frequency & energy of different types of light on the EMR spectrum.
...analyze &/or describe light as both a wave and as a particle (duality).
...relate atomic emission spectra to electrons undergoing atomic absorption and emission.
...state the wavelength range of visible light in nm.
...express electron configurations in orbital box diagrams and noble gas notations (including Cr & Cu exceptions)
...compare and contrast the Bohr model of the atom (orbits) with the wave mechanical model (orbitals).
...visualize and explain orbital shapes (sublevels)
...state possible #'s of electrons in various orbitals, sublevels and principle energy levels.
...determine the number of valence electrons in an atom.
...locate the s,p,d & f blocks on the periodic table.
...explain and diagram major wave vocab (amplitude, speed, wavelength, frequency, Hertz & energy)
...compare/contrast relative speed, wavelength, frequency & energy of different types of light on the EMR spectrum.
...analyze &/or describe light as both a wave and as a particle (duality).
...relate atomic emission spectra to electrons undergoing atomic absorption and emission.
...state the wavelength range of visible light in nm.
| ch_11_review-no_calculations.pptx | |
| File Size: | 5934 kb |
| File Type: | pptx |
| electron_configurations.ppt | |
| File Size: | 1161 kb |
| File Type: | ppt |
| electromagnetic_radiation__light__.ppt | |
| File Size: | 1585 kb |
| File Type: | ppt |
Chapter 3 "I CANs"...
...spell the names and symbols of the first 36 elements.
...identify nonmetals, metals, semi-metals, transition metals, halogens, noble gases, lanthanides, actinides and diatomic elements on the periodic table.
...describe a compounds composition based on its chemical formula.
...identify the three major subatomic particles, their locations and their properties.
...explain and correct Dalton's atomic theory.
...diagram and explain Thomson and Rutherford's experiments.
...interpret and create proper notation for isotopes using both hyphen notation and atomic symbols.
...calculate an element's average atomic mass from isotopic percent abundance data and vice versa.
...properly describe the formulation of ions from their parent atoms.
...predict the neutral compound formed from a cation/anion pair..
...spell the names and symbols of the first 36 elements.
...identify nonmetals, metals, semi-metals, transition metals, halogens, noble gases, lanthanides, actinides and diatomic elements on the periodic table.
...describe a compounds composition based on its chemical formula.
...identify the three major subatomic particles, their locations and their properties.
...explain and correct Dalton's atomic theory.
...diagram and explain Thomson and Rutherford's experiments.
...interpret and create proper notation for isotopes using both hyphen notation and atomic symbols.
...calculate an element's average atomic mass from isotopic percent abundance data and vice versa.
...properly describe the formulation of ions from their parent atoms.
...predict the neutral compound formed from a cation/anion pair..
| chapter_3_flyswatter_vocab_review.docx | |
| File Size: | 14 kb |
| File Type: | docx |
| atomic_history.ppt | |
| File Size: | 2156 kb |
| File Type: | ppt |
| isotopes___ions.pptx | |
| File Size: | 476 kb |
| File Type: | pptx |